Healthcare & Laboratory Labels Market Forecast 2026–2036: Market to Reach USD 11.2 Billion by 2036 at 2.7% CAGR
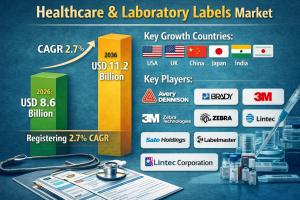
Healthcare & laboratory labels market is anticipated to grow from USD 8.6 billion in 2026 to USD 11.2 billion by 2036, registering a 2.7 % CAGR.
NEW YORK, DE, UNITED STATES, February 23, 2026 /EINPresswire.com/ -- The Healthcare & Laboratory Labels Market is projected to expand from USD 8.6 billion in 2026 to USD 11.2 billion by 2036, registering a 2.7% CAGR. Growth is increasingly shaped by compliance economics, automation density, and rising performance thresholds rather than simple healthcare expansion.
As digitisation deepens across hospitals and laboratories, labels are evolving from consumables into compliance-critical infrastructure embedded within patient safety and diagnostic reliability frameworks.
Healthcare & Laboratory Labels Market Market Snapshot (2026–2036)
• Market size in 2026 USD 8.6 billion
• Market size in 2036 USD 11.2 billion
• CAGR (2026–2036) 2.7%
• Leading end user and share Hospitals – 40.0%
• Key growth countries USA, UK, China, Japan, India
• Core technology shift RFID and advanced 2D codes for non-line-of-sight readability
• Primary growth driver Compliance mandates and automation integration
• Top companies Avery Dennison, Brady, 3M, Zebra Technologies, Sato Holdings, Labelmaster, Lintec Corporation
Market Momentum (2026–2036 Value Path):
The Healthcare & Laboratory Labels Market stands at USD 8.6 billion in 2026, reflecting compliance-driven upgrades and expanding automation across clinical environments. By 2028, procurement increasingly prioritizes validated adhesive durability and non-line-of-sight readability as robotic lab systems scale. Entering 2030, UDI enforcement and direct marking requirements under 21 CFR 801.45 tighten acceptable failure thresholds. By 2031, smart labeling integration with hospital information systems (HIS) and laboratory information systems (LIS) becomes operationally embedded.
In 2033, automation density and intelligent tag deployment further elevate scan reliability as a patient safety control. By 2035, the market approaches its projected USD 11.2 billion valuation by 2036, driven by hardened compliance standards and digitized workflows.
Why the Healthcare & Laboratory Labels Market Is Growing
The Healthcare & Laboratory Labels Market is expanding due to:
• UDI and device identification mandates elevating durability requirements
• Automation reducing tolerance for manual verification
• Increased traceability in rare and complex disease workflows
• Growth in RFID and advanced 2D code adoption
• Adhesive specifications validated for sterilisation, cryogenic storage, and chemical wipe-down cycles
• Rising sustainability expectations aligned with recyclability without compromising scan integrity
Get Access of Report Sample: https://www.futuremarketinsights.com/reports/sample/rep-gb-12535
Segment Spotlight:
1) End User: Hospitals Lead with 40.0%: Hospitals account for 40.0% of market share in 2026, reflecting the highest concentration of risk-bearing workflows. Applications span bedside medication administration, specimen collection, sterile processing, and device reprocessing. Label constructions must withstand sterilisation cycles, chemical exposure, abrasion, and handling stress. Interoperability across printers, scanners, and clinical systems raises switching costs, reinforcing hospital dominance in the Healthcare & Laboratory Labels Market.
2) Diagnostic Laboratories: Automation-Sensitive Growth: Diagnostic labs represent the most automation-sensitive segment. Robotics, conveyors, and enclosed systems demand non-line-of-sight readability and consistent print integrity. Cold storage resistance, centrifugal stress tolerance, and high-speed application reliability define procurement specifications. RFID and advanced 2D codes increasingly enable throughput optimization and error reduction.
3) Pharmaceutical Companies & Research Institutions: Pharmaceutical companies and research institutions purchase labels as part of structured quality systems. Demand centers on chain-of-identity protocols, cold-chain resilience, multilingual compliance, and long-term storage durability. As biologics and gene therapy pipelines expand, controlled labeling frameworks become critical to audit readiness and traceability.
Drivers, Opportunities, Trends, Challenges
• Drivers: Regulatory mandates elevate labeling from packaging detail to compliance-critical infrastructure. The 21 CFR 801.45 direct marking framework increases remediation risk when identification degrades. Adhesive and face stock durability thresholds are resetting minimum performance expectations.
• Opportunities: Smart labeling adoption expands with automation programs. RFID and advanced 2D codes support real-time visibility, multi-site traceability, and audit readiness. Intelligent labels increasingly integrate with data capture platforms and enterprise governance systems.
• Trends: Materials science is emerging as a core competitive advantage. Buyers reduce variability in adhesive chemistry and print permanence to prevent scan failures. Sustainability integration is also accelerating, illustrated by recyclable RFID constructions compatible with PET recycling streams.
• Challenges: Trade policy uncertainty and input volatility moderate full-scale RFID conversion. Tag pricing sensitivity leads to phased adoption, concentrating smart labeling in high-risk workflows first while hybrid barcode environments persist.
Competitive Landscape
• Avery Dennison strengthens positioning in intelligent labels and recyclable RFID systems.
• Brady expands identification solutions through durable marking and specialty capabilities.
• Zebra Technologies operates at the intersection of data capture and workflow digitisation.
• 3M, Sato Holdings, Labelmaster, and Lintec Corporation compete through materials science, printing ecosystems, and healthcare-specific catalogues.
FAQ
How large will the Healthcare & Laboratory Labels Market be by 2036?
The market is projected to reach USD 11.2 billion by 2036.
What is the expected CAGR?
The industry is forecast to grow at 2.7% from 2026 to 2036.
Who is the leading end user?
Hospitals hold the largest share at 40.0% in 2026.
Which countries show strong growth?
Japan (4.5%), India (4.4%), China (4.1%), USA (3.7%), and UK (3.5%).
What is driving smart labeling adoption?
Automation density, regulatory mandates, and the need for non-line-of-sight readability.
Why is materials science critical?
Durability under sterilisation, cryogenic storage, and chemical exposure directly impacts scan integrity and compliance.
Why FMI: https://www.futuremarketinsights.com/why-fmi
Have a Look at Related Research Reports on the Packaging Domain:
Cosmetic Tube Packaging Market: https://www.futuremarketinsights.com/reports/cosmetic-tube-packaging-market
Foam Tape Market: https://www.futuremarketinsights.com/reports/foam-tape-market
Plastic-free Packaging Market: https://www.futuremarketinsights.com/reports/plastic-free-packaging-market
About Future Market Insights (FMI)
Future Market Insights, Inc. (FMI) is an ESOMAR-certified, ISO 9001:2015 market research and consulting organization, trusted by Fortune 500 clients and global enterprises. With operations in the U.S., UK, India, and Dubai, FMI provides data-backed insights and strategic intelligence across 30+ industries and 1200 markets worldwide.
Sudip Saha
Future Market Insights Inc.
+1 347-918-3531
email us here
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.